| Make a google doc and answer the following. Submit it through the form fill for your respective class. Name:___________________ 1. On a scale of 1-10 how much did you and your teamates help each other out. Team Member 1.Name:_________________1-10________ Team Member 2.Name:_________________1-10________ Team Member 3.Name:_________________1-10________ Self Evaluation:_________________1-10________ 2. What were challenges you faced while doing your final information festival? 3. How did you help your team successfully complete your information festival? 4. What did you learn/re-learn? Have a great summer break!!!!! |
|
0 Comments
Your task as a class is to create a fair final examination for another class and they will be creating your final examination. Your final will be on Friday May 19th and is open book, open note, and you can work in a group of 3. We will begin it in class and if you don't finish you can take it home to completed by Monday May 22nd.
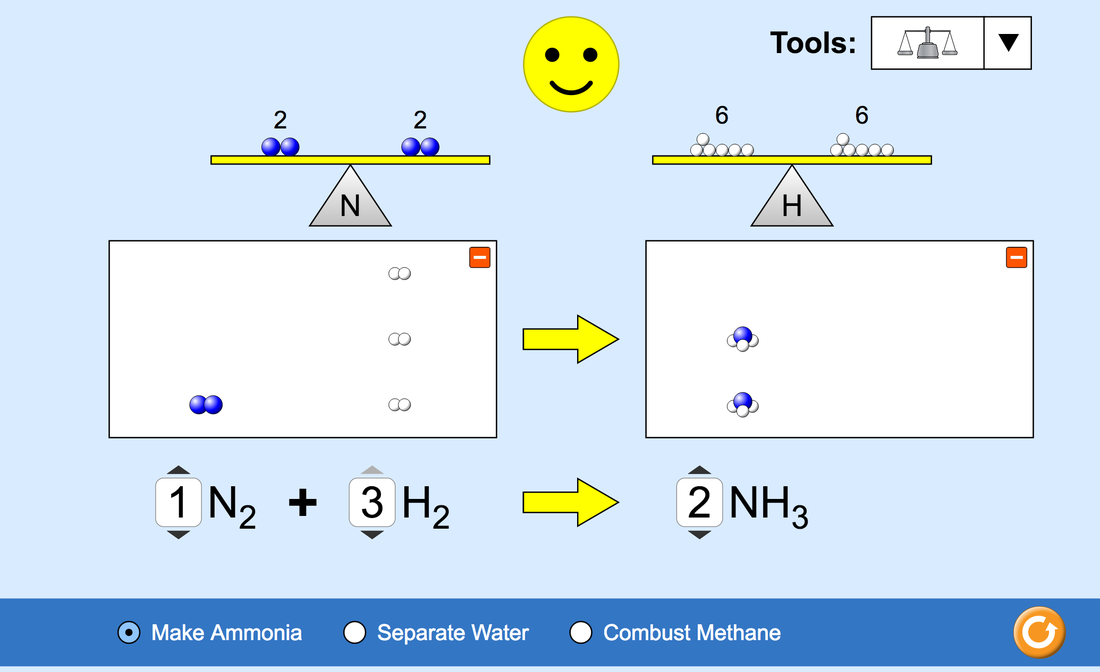
Please enter your questions by subject area into the following documents. Put initials next to your personal questions so I know what you contributed to making the exam. I suggest doing your work on a personal google doc and adding it to the class document when you are done in case someone is immature and tries to delete your work. Chemistry Period 2 Chemistry Period 4 Chemistry Period 5 If anyone deletes or messes with other peoples work they will get an automatic 0!!!! The exam should review the following: Balancing Chemical Equations Nuclear Energy & Alternatives Nuclear Energy: The Pro's, The Con's, and the science behind it. Ex. "Draw a diagram of how a nuclear energy plant works." Radioisotopes Radiometric Dating Half Lives What's an isotope? Flint Water Crisis Solute Concentration: ppm, ppb What are the # of grams in 1 L of water that has 13,000ppb lead, 5,000ppb, 5ppb, and 0 ppb. Graph the safe levels, levels of concern, toxic waste level, and flint. Health affects of contaminants in drinking water and their "safe concentrations." Molarity & Dilutions Molarity of solutions and how to make Dilutions of a liquid. "You may want to make this an active question that they have to perform during class?" Acids & Bases What is an acid/a base? What is the pH scale? If you go from a pH of 8 to a pH of 4, what is the change in Hydrogen Ion concentration? Electronegativity and Bonding Electronegativity Be able to determine if a bond is a Covalent, Polar Covalent, or Ionic Bond using electronegativity values Covalent, Polar Covalent, and Ionic Bonds? How do Polar Covalent bonds cause Hydrogen Bonds.? Did you find any patterns when creating your electronegativity graph? Conductivity Meters How do you make one? What are they used for? What is the difference between using distilled water and water with dissolved salt? What happens if you add an acid? (Think of the penny battery.) Electron Configuration Aufbau principle: core electron: electron configuration Hund’s rule orbital diagram valence electrons valence shell Phases of a Substance Ionic & Covalent Bonding Naming Compounds Scientific Method First Semester 1. Famous Chemists that Influence the Industrial Revolution and Time Line 2. Periodic Table of Elements & Groupings of Elements (Influential Chemists that developed the periodic table and atomic theories.) 3. Drawing and structure of Atoms: Bohr Model, Planetary Model, Lewis Dot Structure 4. Properties of Water 5. SI Units and the Metric System 6. Conversions ex. kg-g, miles-km,..... 7. Calculating Molar Mass and the number of Atoms or Molecules using Avogadros # 8. Accuracy vs. Precision 9. Density 10. Bonding and Types of Molecules or Compounds 11. How a battery works 12. Elephant Toothpaste: reaction of hydrogen peroxide and yeast 13. Oxidation and Reduction Reactions 14. Exothermic & Endothermic Reactions Due on Friday May 12th Do the worksheet by Friday. It was due on Monday but I have extended the deadline to Friday because it was classwork and wasn't on my weebly.
Watch the PBS video from Rare Earth Metals on. The last three chapters of the PBS video that is in the DVD Player. Then Watch Bill Nye Nuclear Energy and Waste In a google slide show or prezi answer the following (Hand in through the form fill): What is an Isotope? What is a half life? How does Bill Nye show the concentrations of radioactive materials over 240,000 years? How can the proportions of isotopes be used to date objects? How are they used in the PBS video? How can we use them to date things in Hawaiʻi? What is a fission reaction? What are some advantages of nuclear energy and what are some disadvantages of nuclear energy? Which elements are used for nuclear energy? How does a nuclear power plant work? (Draw a diagram) What is the current plan to deal with nuclear waste? Is it safe? Where is waste in the U.S. currently slated to be stored? What are alternatives? What are major incidents regarding nuclear energy? When did they happen? Do any have the potential to affect us in Hawaiʻi? Explain What are the moral or ethical ways to move forward in regards to energy? Site your sources! Chernobyl Disaster, Soviet Union 1986
The next two class periods we will be making your exploratorium project/doing your project.
In your notebooks you need to write: Name:_____________ Date:______________ Title:______________ Materials: Methods: Trial x Observations: What are you measuring/looking for? What questions did you end up having? What Next? Please create a blog with photos and an explanation. Submit the link through the google form fill. 20 pts. extra credit
http://mauinow.com/2017/04/20/maui-joins-march-for-science-events-on-earth-day-2017/ 9:00-10:30 on the great lawn at UH Maui College, march down Ka'ahumanu Avenue. Write a 1 page reflection with a photos of your experience. Interview 5 people on why they are marching. Have Fun!!!! https://docs.google.com/spreadsheets/d/1pZQK8mY3u4WHBmzVwb0XYtHhfPwn-6fhClgIW3oi25U/edit#gid=0 What you are assigned to make an info-graphic or video for.
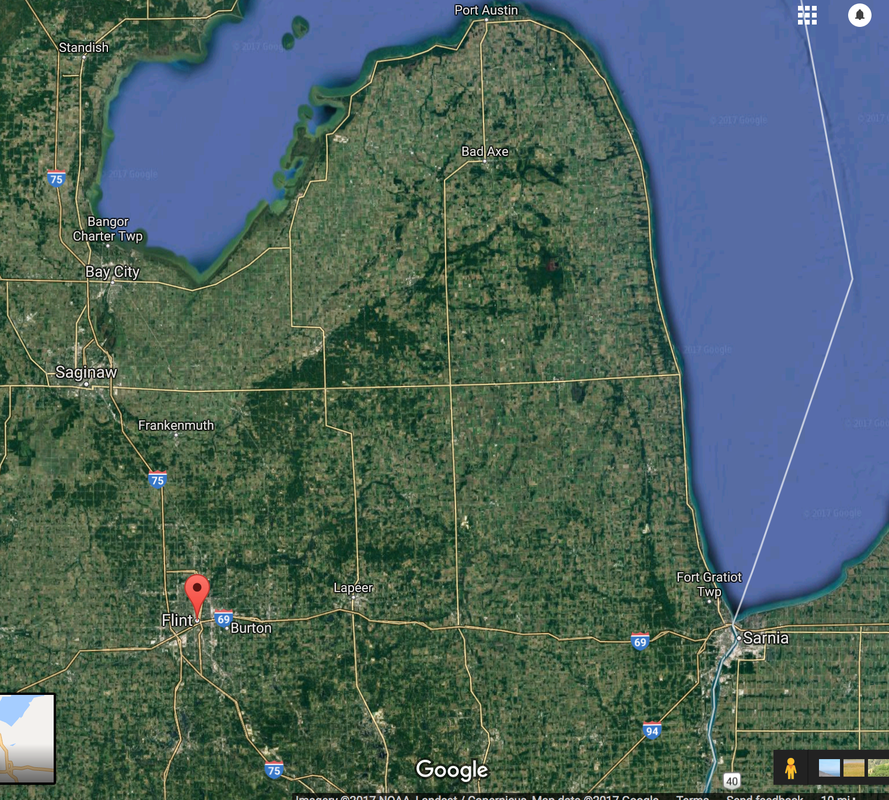
Assignment Create a chronological illustrated narrative about Flint Michigan’s water crisis tracing events as they unfolded. Include any events that contaminated Hawaii's water whether ocean water or drinking water. https://www.tiki-toki.com/ Erin Brockovich on Flint 1. Geography: Illustrate map showing location of Flint Michigan 2. Demographics: Chart population analysis in relation to economy, government assistance, employment, etc. 3. Trace Flint’s water source for many years-Where did it come from? Was there periodic checking for contaminants? Had there ever been a reason to check for contaminants? How often does the EPA run tests? Does the agency have to have a reason? 4. What was the reason behind the change in the water system? Who knew about it? What channels need to be considered before such a decision can be made? 5. Who were the “whistle blowers?” What were the first indicators that something was wrong? At what time was the EPA notified? What would the EPA have to do once proven that a problem exists? What was the first step in testing? How is it done? Who does it? Who gets the results? 6. What did they find when first tested? What was initially done? Was there an increase in the lead levels over time? What were the numbers? Create a chart over time showing results of testing. 7. Why was there a cover-up? Who was involved? What happened to these people? What did the people of Flint want done to these people who were responsible? (Research news articles) 8. Flint today: How was the situation solved? What was done to make water safe for these people? How many people/children have been affected? How are these people being compensated? 9. How was the body affected? Illustrate how lead enters the body and what it does to each organ. 10. Has there ever been a time when Maui’s water was compromised? Research 11. Write a letter to the EPA, the Governor, and the President for the people of Flint. 12. What does it mean when there are so many “parts” of toxins in water? Is there any safe levels? How do you get rid of lead once it is present? 13. What are other toxins that may be in drinking water? What are their sources? What lead to the Flint Michigan Crisis?
What is the crisis? What is happening now? What are the # of grams in 1 L of water that has 13,000ppb lead, 5,000ppb, 5ppb, and 0 ppb. Graph the safe levels, levels of concern, toxic waste level, and flint. Lead Health Affects: https://youtu.be/hqnaq2Vn-CA EPA's thoughts about lead in tap water & medical conditions as a result of lead in tap water. What are considered the "safe" levels of lead in tap water? As a class Watch the video on Water and Solutions: Take notes on the video. The video is pretty fast so when they are doing calculations in the video try to pause it or replay the calculations so we can take notes.
Do the Molarity Calculation Practice Problems on Khan Academy https://www.khanacademy.org/science/chemistry/states-of-matter-and-intermolecular-forces/mixtures-and-solutions/e/molarity-calculations-exercise Do the End of Chapter Exercises on Molarity and Dillutions at the bottom of the section that you annotated. https://cnx.org/contents/[email protected]:41l9H_NI@8/Molarity When you are done; annotate 6.4 Other Units for Solution Concentrations. This will be do on Wednesday. Molarity Simulation shows the relationship of the solute concentration to the amount of solvent.
Read and Annotate the Chapter on Molarity and Dilutions due on Monday 6.3 Molarity Link |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2017
Categories |
||||||




 RSS Feed
RSS Feed
