There will be an information festival on Monday Dec. 19th. You are allowed an 8.5 X 11 inch (21.59 cm x 27.94 cm) hand written "reference sheet" that you will hand in with your information festival for 5 points. You must make the reference sheet your self, you can't increase surface area by any means, and you can't zerox a friends reference sheet. Material from Quarter 2 will be the focus but you should know material from quarter one.
Quarter 2
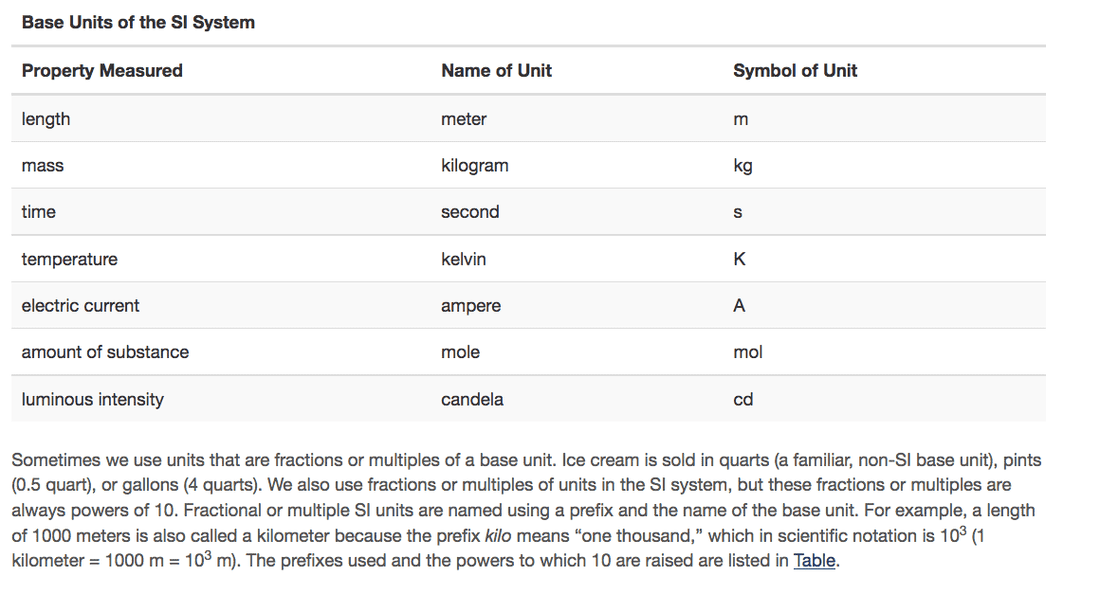
SI Units (Metric System) Be able to identify the property measured with the appropriate units. Indicate the SI base units or derived units that are appropriate for the following measurements:
(a) the length of a marathon race (26 miles 385 yards)
(b) the mass of an automobile
(c) the volume of an olympic swimming pool
(d) the speed of an airplane
(e) the density of gold
(f) the area of a football field
(g) the maximum temperature at the South Pole on April 1, 1913
Indicate the SI base units or derived units that are appropriate for the following measurements:
(a) the mass of the moon
(b) the distance from Dallas to Oklahoma City
(c) the speed of sound
(d) the density of air
(e) the temperature at which alcohol boils
(f) the area of the state of Delaware
(g) the volume of a flu shot or a measles vaccination
Give the name of the prefix and the quantity indicated by the following symbols that are used with SI base units. Ex. c=centi
(a) c
(b) d
(c) G
(d) k
(e) m
(f) n
(g) p
(h) T
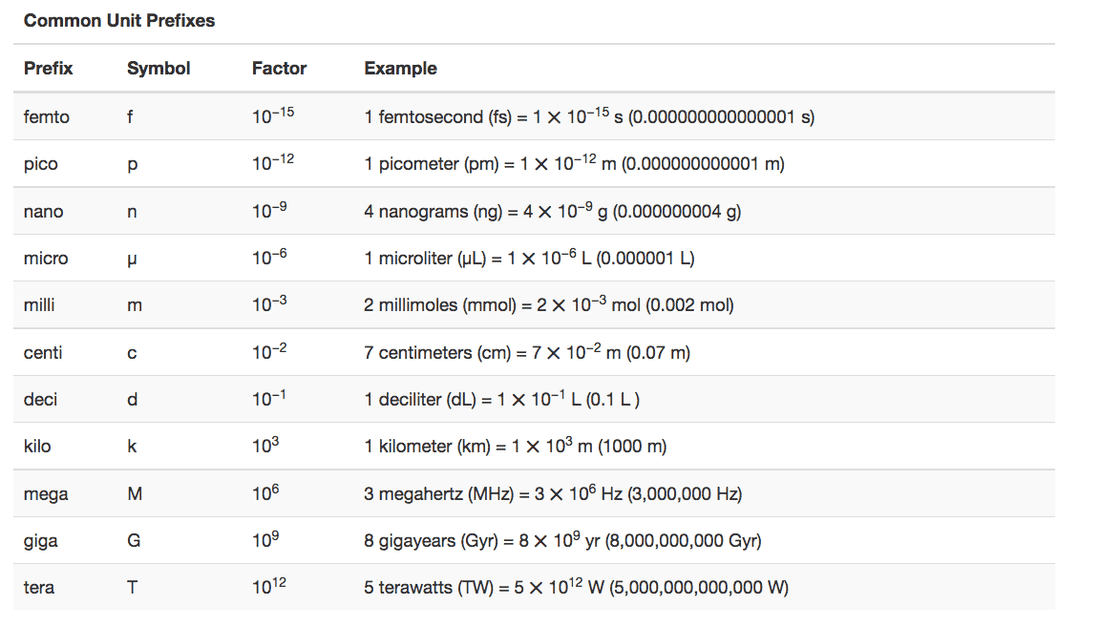
Give the name and symbol of the prefixes used with SI units to indicate multiplication by the following exact quantities.
(a) 103
(b) 10−2
(c) 0.1
(d) 10−3
(e) 1,000,000
(f) 0.000001
Check out the scale of the universe to get a good understanding of what the prefixes mean to the size of objects you can relate to.
Quarter 2
SI Units (Metric System) Be able to identify the property measured with the appropriate units. Indicate the SI base units or derived units that are appropriate for the following measurements:
(a) the length of a marathon race (26 miles 385 yards)
(b) the mass of an automobile
(c) the volume of an olympic swimming pool
(d) the speed of an airplane
(e) the density of gold
(f) the area of a football field
(g) the maximum temperature at the South Pole on April 1, 1913
Indicate the SI base units or derived units that are appropriate for the following measurements:
(a) the mass of the moon
(b) the distance from Dallas to Oklahoma City
(c) the speed of sound
(d) the density of air
(e) the temperature at which alcohol boils
(f) the area of the state of Delaware
(g) the volume of a flu shot or a measles vaccination
Give the name of the prefix and the quantity indicated by the following symbols that are used with SI base units. Ex. c=centi
(a) c
(b) d
(c) G
(d) k
(e) m
(f) n
(g) p
(h) T
Give the name and symbol of the prefixes used with SI units to indicate multiplication by the following exact quantities.
(a) 103
(b) 10−2
(c) 0.1
(d) 10−3
(e) 1,000,000
(f) 0.000001
Check out the scale of the universe to get a good understanding of what the prefixes mean to the size of objects you can relate to.
Conversions Moles and Avagodro's Number
The F-35 has a maximum speed of Mach 1.6, which is 1.6 times the speed of sound, or about 1,200 mph. What is the maximum speed of the F-35 jet in km?
What is Avogadro's Number
What is a mole?
How do you find how much a mole of an element weighs?
How do you find how much a mole of a molecule weighs?
What does Avogadro's Number tell you?
How many atoms are in a mole of copper?
How many moles are in 254.2 g of copper?
How many atoms are in 254.2 g of copper?
How many moles of copper does 2.4088 X 10^24 atoms make up?
How many grams of copper does 2.4088 X 10^24 atoms make up?
Accuracy vs. Precision
Precision: Measurements are said to be precise if they yield very similar results when repeated in the same manner.
Accuracy: A measurement is considered accurate if it yields a result that is very close to the true or accepted value.
The F-35 has a maximum speed of Mach 1.6, which is 1.6 times the speed of sound, or about 1,200 mph. What is the maximum speed of the F-35 jet in km?
What is Avogadro's Number
What is a mole?
How do you find how much a mole of an element weighs?
How do you find how much a mole of a molecule weighs?
What does Avogadro's Number tell you?
How many atoms are in a mole of copper?
How many moles are in 254.2 g of copper?
How many atoms are in 254.2 g of copper?
How many moles of copper does 2.4088 X 10^24 atoms make up?
How many grams of copper does 2.4088 X 10^24 atoms make up?
Accuracy vs. Precision
Precision: Measurements are said to be precise if they yield very similar results when repeated in the same manner.
Accuracy: A measurement is considered accurate if it yields a result that is very close to the true or accepted value.
If you were a Maui County Council Member, whould you pass the Polystyrene Ban? Explain your rational! What are the pro's what are the cons? Do the pro's outweigh the cons?
What are the effects this would have to the consumers and to the restaurants financially?
Are there alternatives to polystyrene containers?
What are the effects on the oceans if this is not passed?
Why is styrofoam and single use plastics bad for our oceans?
How long does it take for styrofoam and plastic to degrade?
What is the molecular formula of polystyrene?
How much does one mole of polystyrene weigh?
How many molecules are in 85 grams of polystyrene?
What are the effects this would have to the consumers and to the restaurants financially?
Are there alternatives to polystyrene containers?
What are the effects on the oceans if this is not passed?
Why is styrofoam and single use plastics bad for our oceans?
How long does it take for styrofoam and plastic to degrade?
What is the molecular formula of polystyrene?
How much does one mole of polystyrene weigh?
How many molecules are in 85 grams of polystyrene?
Density
What are factors that change the volume of a substance?
What are factors that change the density of water?
What is the formula for density?
What is the formula for density?
What is the density of a cube that is 25 cm X 10 cm X 12 cm and weighs 45 g?
What is the density of a marble that causes the water in a graduated cylinder to go from 110 ml to 150 ml and has a weight of 70 grams?
Which liquid is less dense and would float on top, fresh water or salt water?
See images bellow!!!!! Here is the link to the chapter if you need it.
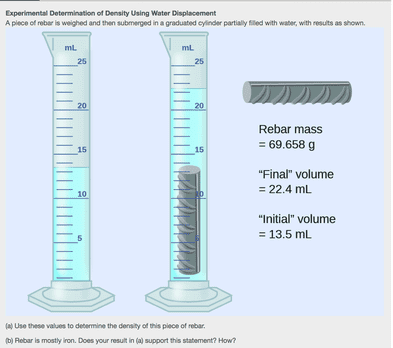
Experimental Determination of Density Using Water Displacement A piece of rebar is weighed and then submerged in a graduated cylinder partially filled with water, with results as shown. (a) Use these values to determine the density of this piece of rebar.
(b) Rebar is mostly iron. Does your result in (a) support this statement? How?
SolutionThe volume of the piece of rebar is equal to the volume of the water displaced:
volume=22.4 mL−13.5 mL=8.9 mL=8.9 cm3(rounded to the nearest 0.1 mL, per the rule for addition and subtraction)
The density is the mass-to-volume ratio:
density=mass / volume=69.658 g / 8.9 cm3=7.8 g/cm3(rounded to two significant figures, per the rule for multiplication and division)
Check Your Learning An irregularly shaped piece of a shiny yellowish material is weighed and then submerged in a graduated cylinder, with results as shown.
(a) Use these values to determine the density of this material.
(b) Do you have any reasonable guesses as to the identity of this material? Explain your reasoning.
What are factors that change the volume of a substance?
What are factors that change the density of water?
What is the formula for density?
What is the formula for density?
What is the density of a cube that is 25 cm X 10 cm X 12 cm and weighs 45 g?
What is the density of a marble that causes the water in a graduated cylinder to go from 110 ml to 150 ml and has a weight of 70 grams?
Which liquid is less dense and would float on top, fresh water or salt water?
See images bellow!!!!! Here is the link to the chapter if you need it.
Experimental Determination of Density Using Water Displacement A piece of rebar is weighed and then submerged in a graduated cylinder partially filled with water, with results as shown. (a) Use these values to determine the density of this piece of rebar.
(b) Rebar is mostly iron. Does your result in (a) support this statement? How?
SolutionThe volume of the piece of rebar is equal to the volume of the water displaced:
volume=22.4 mL−13.5 mL=8.9 mL=8.9 cm3(rounded to the nearest 0.1 mL, per the rule for addition and subtraction)
The density is the mass-to-volume ratio:
density=mass / volume=69.658 g / 8.9 cm3=7.8 g/cm3(rounded to two significant figures, per the rule for multiplication and division)
Check Your Learning An irregularly shaped piece of a shiny yellowish material is weighed and then submerged in a graduated cylinder, with results as shown.
(a) Use these values to determine the density of this material.
(b) Do you have any reasonable guesses as to the identity of this material? Explain your reasoning.

Check Your Learning An irregularly shaped piece of a shiny yellowish material is weighed and then submerged in a graduated cylinder, with results as shown.
(a) Use these values to determine the density of this material.
(b) Do you have any reasonable guesses as to the identity of this material? Explain your reasoning.
(a) Use these values to determine the density of this material.
(b) Do you have any reasonable guesses as to the identity of this material? Explain your reasoning.
Important Q 1 Vocab Words
Chemistry Quarter 1 Information Festival Material
Big Bang Theory
Matter
Mass
Atom
Atomic #
Atomic Mass
Atomic Particles: be able to make a table with the atomic mass unit of each particle, their charges, and where they are found within an atom
Elements
Atom
Proton
Neutron
Electron
Varying types of an Element
Isotope
Cation
Anion
Octet Rule
Valence Shell
Periodic Table
elements that we used this quarter
Hydrogen
Oxygen
Nitrogen
Carbon
Sodium Chlorine
Iron
Zinc
Copper
Groupings of elements in the periodic table
Alkali Metal
Alkaline Earth Metals
Transition Metal
Basic Metal
Semimetal
Nonmetal
Halogen
Lanthanides Actinide
Nobel Gas
Types of Bonds: how they are different and be able to draw examples, which one is the strongest and which one is the weakest?
Covalent
Ionic
Hydrogen
Electrostatic Attraction
Compound vs. Molecule
Bohr Model
Planetary Model
Oxidation and Reduction Reaction
-example within the penny battery experiment
Be able to explain how the penny battery worked
Elephant Toothpaste reactants and products (in class or Jimmy Kimmel elephant toothpaste)
Catalyst
Density
How to calculate the Molar Mass of a compound or molecule
Properties of Water
adhesion
cohesion
surface tension
thermal properties: freezing point, melting point, boiling point
density of water at different temperatures
density of water with salt dissolved
universal solvent
conductive properties of water vs table salt dissolved in water
acid
base
Chemistry Quarter 1 Information Festival Material
Big Bang Theory
Matter
Mass
Atom
Atomic #
Atomic Mass
Atomic Particles: be able to make a table with the atomic mass unit of each particle, their charges, and where they are found within an atom
Elements
Atom
Proton
Neutron
Electron
Varying types of an Element
Isotope
Cation
Anion
Octet Rule
Valence Shell
Periodic Table
elements that we used this quarter
Hydrogen
Oxygen
Nitrogen
Carbon
Sodium Chlorine
Iron
Zinc
Copper
Groupings of elements in the periodic table
Alkali Metal
Alkaline Earth Metals
Transition Metal
Basic Metal
Semimetal
Nonmetal
Halogen
Lanthanides Actinide
Nobel Gas
Types of Bonds: how they are different and be able to draw examples, which one is the strongest and which one is the weakest?
Covalent
Ionic
Hydrogen
Electrostatic Attraction
Compound vs. Molecule
Bohr Model
Planetary Model
Oxidation and Reduction Reaction
-example within the penny battery experiment
Be able to explain how the penny battery worked
Elephant Toothpaste reactants and products (in class or Jimmy Kimmel elephant toothpaste)
Catalyst
Density
How to calculate the Molar Mass of a compound or molecule
Properties of Water
adhesion
cohesion
surface tension
thermal properties: freezing point, melting point, boiling point
density of water at different temperatures
density of water with salt dissolved
universal solvent
conductive properties of water vs table salt dissolved in water
acid
base


 RSS Feed
RSS Feed
