| molecule-shapes-student-handout.pdf |
|
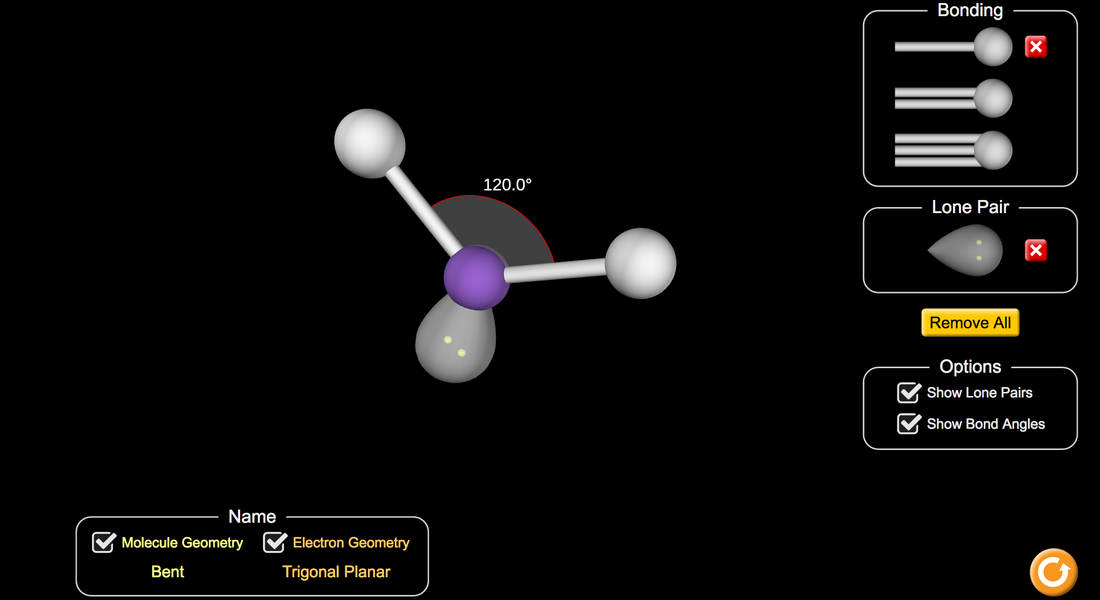
Go to the molecular shape simulation and answer the worksheet questions. You can work in groups of 3 but must have your own worksheet.
0 Comments
In quarter four, we will end our chemistry relationship by building a "exploratorium" exhibit where you are tasked to teach a fellow student, a parent, or a teacher something about chemistry or science in a hands on way.
Today I want you to research hands on activities that engage our curiosity and inquiry into learning more about science and the unknowns. Submit your document through the homework submission form fill found on my home page. What you will hand in: A link describing the proposed exhibit, how does this pertain to chemistry or the inquiry of science, what supplies or tools do you need in order to build it, how can you personalize it as a KCS exhibit? Reflection of your exploratorium exhibit search. Look up and write down the key term definitions in the acid & base houndout that you were given in class.
During class you will work on making your "reference sheet" for the information festival on Friday. Make sure you place all definitions and how to do pertinent problems on your reference sheet. I will give you a periodic table and a electronegativity table. You need to be able to add the orbitals to these tables on your own in order to figure out the electron configuration and to draw an orbital diagram.
Your ea will check to see how much of your cheat sheet you have done. If you didn't do anything you will get no participation points. If you have 1/4 of a page you will get 1.5 points, if you have greater than half a page you will get full credit. I encourage you to use your time wisely. There will be an information festival on Friday March 3rd. You are allowed an 8.5 X 11 inch (21.59 cm x 27.94 cm) hand written "reference sheet" that you will hand in with your information festival for 5 points. You must make the reference sheet your self, you can't increase surface area by any means, and you can't zerox a friends reference sheet. Material from Quarter 2 will be the focus but you should know material from quarter one.
What will we be celebrating: Electronegativity Be able to determine if a bond is a Covalent, Polar Covalent, or Ionic Bond using electronegativity values Covalent, Polar Covalent, and Ionic Bonds? How do Polar Covalent bonds cause Hydrogen Bonds.? Did you find any patterns when creating your electronegativity graph? Covalent Bonding http://mrfitzkcs.weebly.com/chemistry-homework/covalent-bonding-naming-compounds-due-feb-1st Understand the potential energy vs. distance of atoms in a bond. Where will you find the distance of two atoms from one another in a covalent bond? http://hawaii.pbslearningmedia.org/resource/lsps07.sci.phys.matter.covalentbond/covalent-bonding/ In order from weakest to strongest covalent bonds draw a covalent, double covalent, and triple covalent bond. Ionic Bonding Do the following online activity: http://hawaii.pbslearningmedia.org/asset/lsps07_int_ionicbonding/ Electron Configurations and Orbital Diagrams http://mrfitzkcs.weebly.com/chemistry-homework/february-13th-2017 How can we make a cheap accurate and fairly precise conductivity meter? We need a conductivity meter but only have $5 to spend on each conductivity meter. The cheapest one I have found is $22. What is a conductivity meter? How can we make one? What are they used for in chemistry, biology, or oceanography? What makes certain conductivity meters better? Design and source materials to create your own conductivity meter. Open Stax Electron Configurations: https://cnx.org/contents/[email protected]:78vb2wgK@4/Electronic-Structure-of-Atoms-
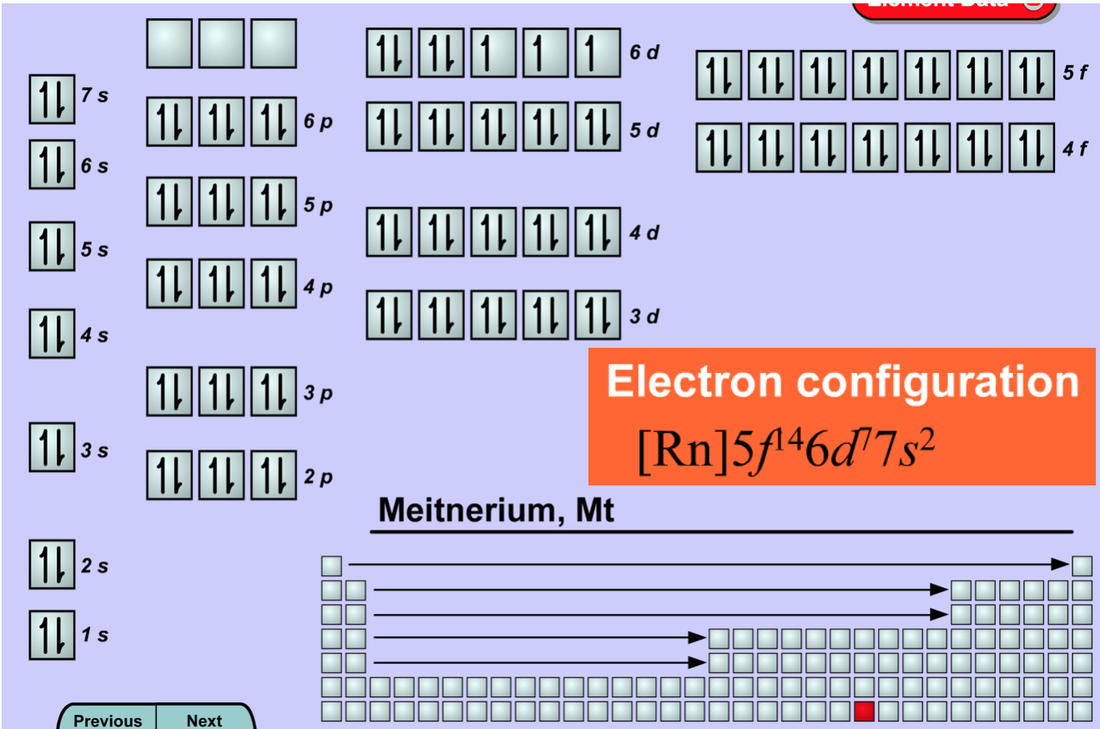
In your notebook answer the end of chapter questions. Make sure you answer them in complete sentences or write the question down first. Define the following: Aufbau principle: core electron: electron configuration Hund’s rule orbital diagram valence electrons valence shell http://hawaii.pbslearningmedia.org/asset/lsps07_int_graphperiodic/
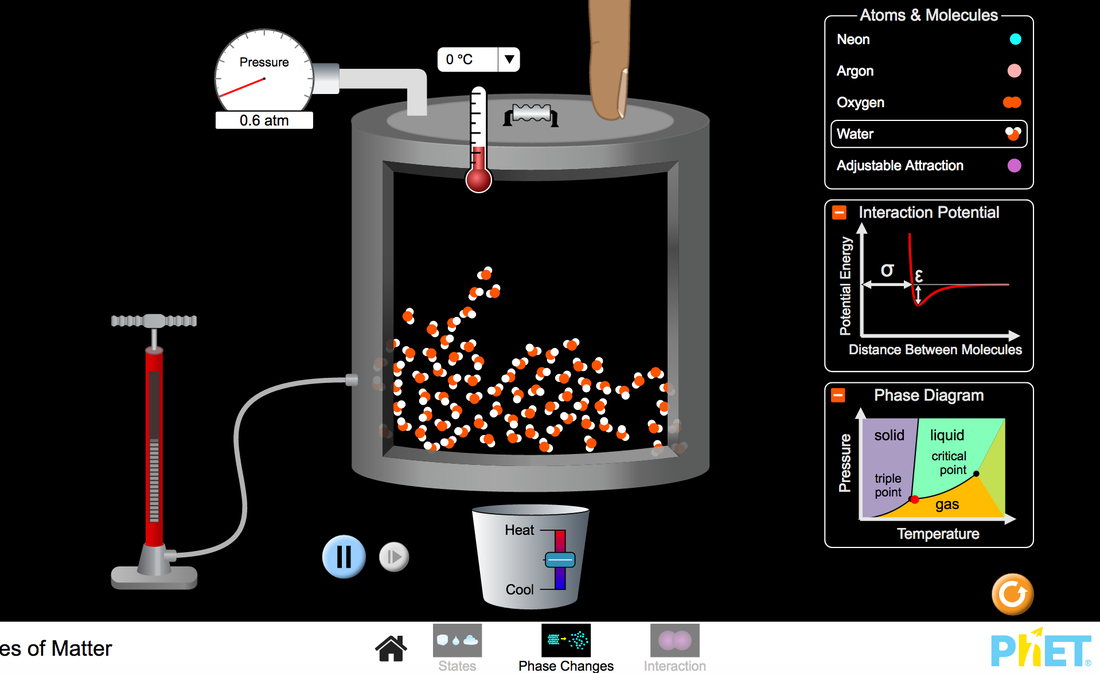
1. Which columns or groups of the periodic table are the s1 Group? The S2 Group? The P1 Group? The 2P2? The P3? The P4? The P5? The P6? The D and the F electron configuration? Which types of elements in the periodic table make up the d & f electron configurations? Without looking try to write the electron configuration for H, He, Li, O, C, Na, Cl, Fe, & No You can click on Element Data to check your work. Phet Diagram: https://phet.colorado.edu/sims/html/states-of-matter/latest/states-of-matter_en.html
Challenge Prompts: Answer the following in your note book. You must either rewrite the question of paraphrase the question in your notebook. • Draw a picture of each substance as a solid, liquid, and gas. Explain the differences and similarities between each state. • Draw a picture of each substance as a solid. Describe how solid water compares to the other substances, and explain why ice floats on water. • Describe a procedure to change the phase of one of the substances. • Explain how a change in temperature affects the pressure inside a container. • Predict what happens to the speed of the atoms and the amount of space between them if (a) heat is added to the system, (b) heat is removed from the system, or (c) the volume of the container is reduced. • Describe how attractive and repulsive forces influence the attraction between two atoms. • Explain the relationship between the attractive forces between atoms and the potential energy graph for the atom pair. Answer the following in your notebooks. You must write the question above your answer or answer the question in a complete sentence when applicable.
Do the following online activity: http://hawaii.pbslearningmedia.org/asset/lsps07_int_ionicbonding/ 1. What is an Ionic Bond? Draw an example 2. What happens as you try to put the two anions (negatively charged ions) together? 3. What happens as you slowly move the oppositely charged ions (cation +, anion -) together? 4. Which atoms in the periodic table tend to form cations? (you can be general in your answer) 5. Which atoms in the periodic table tend to form anions? (you can be general in your answer) 6. What is the ratio of Na cations to Cl anions in a salt crystal? 7. How many Calcium Ions and How many Fluoride ions are in calcium fluoride? 8. What is the ratio? 9. Why do ionic bonds form? 10. What does electronegativity mean? 11. How does the difference in electronegativity determine if a bond is covalent, polar covalent, and ionic? |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2017
Categories |


 RSS Feed
RSS Feed
