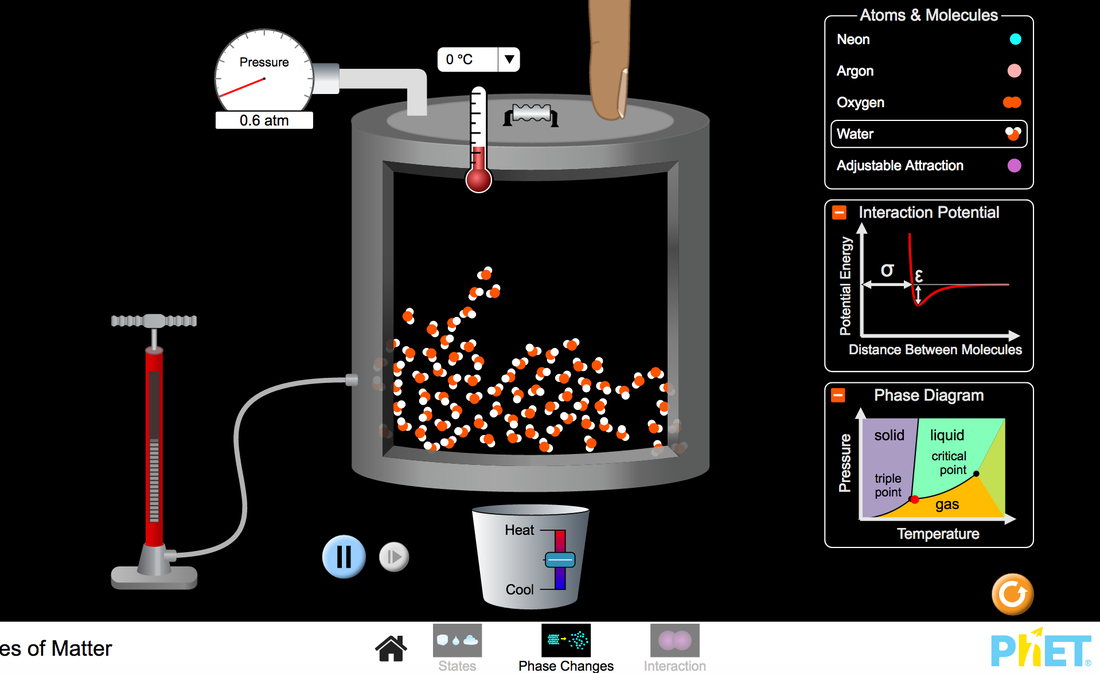
Phet Diagram: https://phet.colorado.edu/sims/html/states-of-matter/latest/states-of-matter_en.html
Challenge Prompts: Answer the following in your note book. You must either rewrite the question of paraphrase the question in your notebook.
• Draw a picture of each substance as a solid, liquid, and gas. Explain the differences and
similarities between each state.
• Draw a picture of each substance as a solid. Describe how solid water compares to the other
substances, and explain why ice floats on water.
• Describe a procedure to change the phase of one of the substances.
• Explain how a change in temperature affects the pressure inside a container.
• Predict what happens to the speed of the atoms and the amount of space between them if (a)
heat is added to the system, (b) heat is removed from the system, or (c) the volume of the
container is reduced.
• Describe how attractive and repulsive forces influence the attraction between two atoms.
• Explain the relationship between the attractive forces between atoms and the potential energy
graph for the atom pair.
Challenge Prompts: Answer the following in your note book. You must either rewrite the question of paraphrase the question in your notebook.
• Draw a picture of each substance as a solid, liquid, and gas. Explain the differences and
similarities between each state.
• Draw a picture of each substance as a solid. Describe how solid water compares to the other
substances, and explain why ice floats on water.
• Describe a procedure to change the phase of one of the substances.
• Explain how a change in temperature affects the pressure inside a container.
• Predict what happens to the speed of the atoms and the amount of space between them if (a)
heat is added to the system, (b) heat is removed from the system, or (c) the volume of the
container is reduced.
• Describe how attractive and repulsive forces influence the attraction between two atoms.
• Explain the relationship between the attractive forces between atoms and the potential energy
graph for the atom pair.
 RSS Feed
RSS Feed
